Reptiles and birds have significant differences in their integument and associated structures compared with mammals, such as scales and feathers. They also have unique infectious and noninfectious dermatologic diseases that are important for veterinarians who treat these species to know. These taxa also have their own specific dermatologic conditions and treatments.
This article highlights significant anatomic differences and unique disease presentations in zoological companion
animal dermatology.
Reptiles
Reptile integument serves many different functions compared to the typical mammalian integument. It is important for water conservation, particularly for desert-dwelling species, and it can play a role in social interaction via color change in certain species, most notably chameleons.
The most significant difference between the reptile integument and mammals is the presence of scales within the stratum corneum. These are comprised of two types of keratin: beta-keratin, which forms the hard scales, and alpha-keratin, which forms the softer connective tissues or “sutures” between the scales. Osteoderms are boney plates found in the dermis in chelonians and crocodilians. The carapace (upper shell) and plastron (lower shell) of chelonians are formed by osteoderms fused with the vertebrae, ribs, and sternum. Coloration of the reptile integument is due to chromatophores and melanocytes. Stacking of these cells on top of one another is present in species that display color change (i.e. chameleons). Reptile skin is relatively aglandular, but there are also species-specific glands.
Almost all reptiles undergo ecdysis (shedding of their skin), which occurs every 10-14 days, on average, for most snakes and lizards. Two patterns of shedding exist, complete (snakes, some lizards), where the entirety of the skin is shed in one piece, and partial (most lizards, chelonians, and crocodilians), where pieces of skin throughout the body are shed at any one time. This process is controlled by thyroid hormones, and have two primary phases: resting (normal appearance of skin) and renewal (shedding).
Disease presentations
Treatment of thermal burns is dependent on severity of lesions, keeping in mind they will likely get worse before improvement is noted. Analgesia is of utmost importance, and there are several recent studies which have proven acceptable analgesics in chelonian and lizard species.
Topical therapies and bandaging can be challenging, especially in snakes. If the burn(s) are severe, parenteral antibiotics (ideally based on a culture and sensitivity) should be considered.
Maintaining a clean environment and minimizing substrate in the enclosure is also of utmost importance. Certain burns may cause permanent scale damage, even after subsequent sheds, which can lead to future dysecdysis.
- Several fungal dermatopathies have been recently described in a variety of reptile species, both captive and wild. Nannizziopsis guarroi (formally called “CANV,” or Chrysosporium anamorph of Nannizziopsis vriesii) was originally referred to as “yellow fungus disease” of mainly bearded dragons, due to the yellow discoloration of the scales in many infected animals.Ophidiomyces ophiodiicola is an important cause of morbidity and mortality in mainly wild snakes,and is also called “Snake fungal disease.” A novel fungus, Emydomyces testavorans, was recently isolated from necrotic shell lesions of freshwater turtles, and may explain why many cases of erosive shell necrosis (sometimes called “shell rot”) were seemingly resistant to treatment with topical and systemic antibiotics.
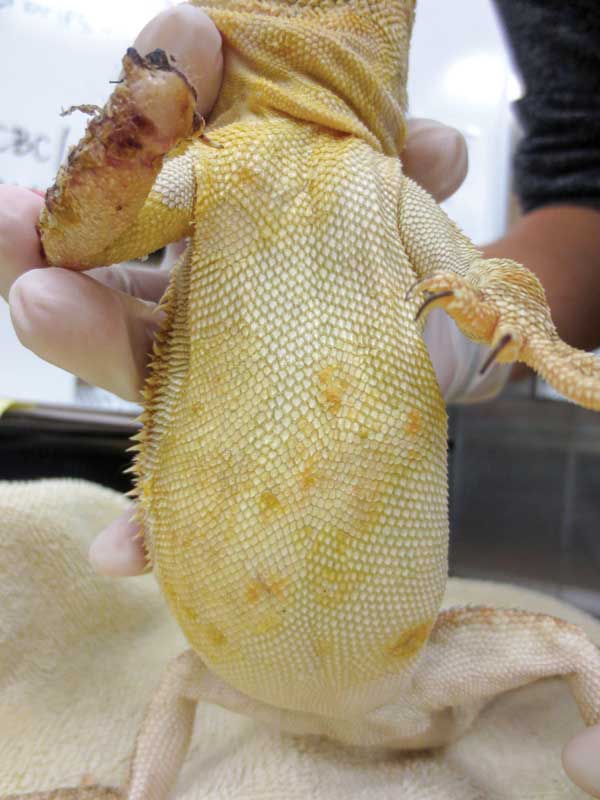
Of these fungal infections, N. guarroi is the most likely encountered in small animal general practice due to the recent and increasing popularity of pet bearded dragons. Clinical signs of N. guarroi infection are dermal erosions, ulcerations, and/or crust formation (Figure 2).
Figure 2. The ventral coleom of a bearded dragon infected with Nannizziopsis guarroi with the classic presentation of yellow-colored multifocal dermatitis. This patient also had several severe ulcerative lesions of all digits of the right forelimb, which lead to amputation of those infected digits. Photo courtesy Dr. Olivia Petritz One cannot visually differentiate bacterial from fungal dermatitis. Crusts or dermal biopsies can be submitted for culture, but this fungus is challenging to grow in vitro, as it requires lower temperatures. Histopathology and PCR techniques are also available. Parenteral antifungal medications are often required for severe infections, and voriconazole is preferred over itraconazole due to less systemic side effects. Successful treatment often requires long treatment periods (> 60 days). Terbinafine has recently been evaluated for treatment of this disease, with successful results—this drug is also significantly cheaper than voriconazole, even when compounded.
- Ophionyssus natricis, often called the snake mite, is the most common mite of reptile species, and it can infect a variety of snakes and lizards. These mites have been documented to transmit several bacteria and viruses. Clinical signs of infection include pruritus/excessive rubbing, dermatitis, dysecdysis.Severe infestations, especially in smaller reptiles, can lead to severe anemia. The mite has a long lifecycle (approximately 40 days), and a significant portion of that is spent off the host in the environment. Therefore, the duration of treatment must be at least 40 days.
Insecticides (all potentially neurotoxic), including ivermectin parenterally and/or topical sprays for the environment (not in chelonians), or topical fipronil have been used for treatment. In addition, predatory mites (Cheyletus eruditus) have also been successfully employed for treatment in a variety of species. This mite is considered a zoonotic disease, and proper precautions should be taken by anyone who handles the infected reptile or its environment.

Birds
True glands, including sweat glands, are absent throughout most of the avian integument. The uropygial (preen) gland is the main sebaceous gland at the base of the tail, but it is not present in all species. Feathers grow from a follicle in the dermis, and the dermal papilla provides blood and nervous supply. One of the main differences between a feather and a mammalian hair is the feather follicle contains a vascular core of dermis as well as epidermis. This is why a feather may bleed profusely when plucked out and why blood feathers should not be cut when wing clipping.
There are four basic types of feathers (See: “Feather terminology”):
- Contour feathers are the outermost “main” feathers. These include the remiges (flight weathers of the wings) and retrices (tail feathers)
- Semipulme feathers are used for insulation
- Filoplumes are highly innervated feathers found near each contour feather follicles are equivalent to mammalian whiskers, bristle feathers have no barbs and are found around the eyes of some avian species (often mistaken for eyelashes, which no bird possesses)
- Down feathers, which are used for insulationA molt is the replacement and shedding of old feathers. Avian species molt at different times and frequencies during the year. It usually follows a symmetric and distinct pattern: primary flight feathers then secondary flight feathers, followed by body feathers. Molt is triggered by many factors, including thyroid hormones, photoperiod, and temperature.
Disease presentations - Feather destructive behavior (FDB), also known as feather damaging behavior or feather picking, affects an estimated 10-15 percent of captive psittacines worldwide. Damage is always restricted to the areas the bird can reach with its beak, which excludes its head. This physical exam finding (pristine and intact head feathers) is important to differentiate FDB from other medical causes of feather loss, including some highly infectious and fatal viral diseases. This behavior can progress to self-trauma of the underlying skin and musculature, which is referred to as self-mutilation or automutilation (Figure 3).
Clinical signs of FDB can vary from mild localized feather damage to complete removal of all feathers (including underlying downy feathers) and soft tissue damage. Removal of the feathers is more common on the chest, ventral wings, and medial thighs—chewing of feathers is more common on the primary flight feathers (remiges) and tail feathers (retrices).
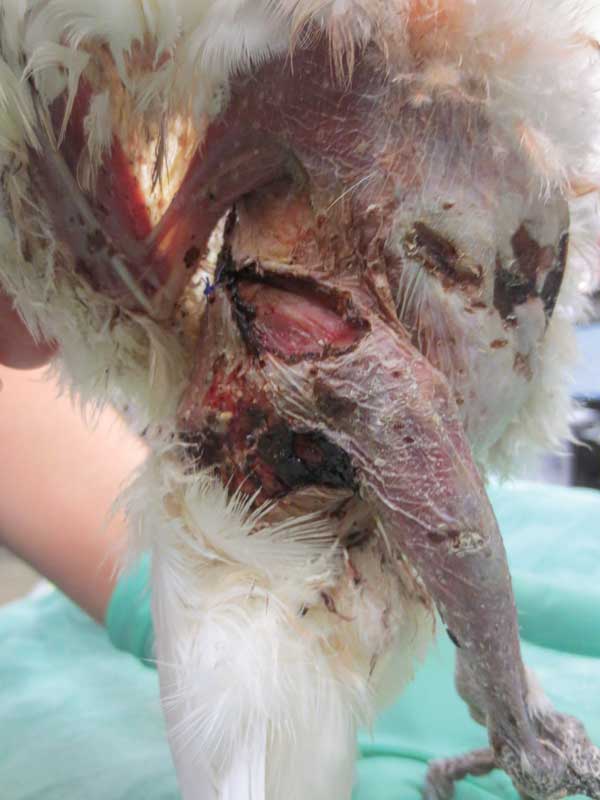
Figure 3. Significant self-trauma with full thickness skin defects caused by automutilation in a Goffin’s cockatoo that was singly housed. Photo courtesy Dr. Olivia Petritz African grey parrots and cockatoos appear to be the most at-risk species, and macaws, Eclectus parrots, Quaker parrots, and conures are also commonly affected. Amazon parrots, cockatiels, and budgies uncommonly present with FDB.
FDB is a multifactorial behavioral disorder, with medical, genetic, psychologic, neurobiologic, and socioenvironmental factors all contributing to disease. Numerous underlying medical conditions have been hypothesized to stimulate this behavior, but no reliable relationships have been definitively proven.
If feather destructive behavior is confined to a localized area (such as over a joint, or the synsacrum, where the kidneys are located), an underlying disease in that particular region or organ system may be present and the cause for the feather damaging behavior.
Other risk factors include nutritional deficiencies and dietary imbalances, airborne and topical toxins/irritants, and low environmental humidity.
Lack of social interaction, lack of a sexual partner/mate (~99 percent of all parrots are not spayed or neutered), lack of locomotor activities, and lack of foraging opportunities have all been theorized to contribute to FDB, but only lack of foraging has been scientifically proven to be associated with FDB.
In addition, the presence of adverse stimuli (abnormal photoperiod, lack of sleep, overcrowding, sudden environmental changes) may lead to chronic stress and subsequent FDB. In this theory, FDB develops as a coping mechanism for these stressors, and serves as a method of tension release.
A complete and thorough history should be taken, including a complete behavioral history to help identify any triggers or stimuli for the behavior. Sadly, many parrots have unknown or incomplete full histories due to serial ownership during their long lifespans.
Any medical issue identified should be treated as indicated, most commonly a bacterial or fungal dermatitis, which could be secondary to FDB as a chronic behavioral condition. Promoting a more stimulating environment is one of the main components of any treatment plan to reduce FDB.
Immediate placement of an E-collar is always recommended for cases of self-mutilation until the underlying skin can heal appropriately and changes are made to help mitigate future self-injurious behaviors.
Pharmacologic treatments used for FDB have included anxiolytic drugs, antipsychotic drugs, tricyclic antidepressants, selective serotonin reuptake inhibitors, and opioid antagonists—all with varying degrees of long-term success. There are several reported cases of neurologic signs and death in parrots following a short course of TCAs for FDB, specifically clomipramine. Unfortunately, there is very limited pharmacokinetic, pharmacodynamic, and safety data in any parrot species for many of these drugs used in the treatment of FDB.
Drug therapy should never be used as a solo treatment method for FDB. Due to the chronicity of the behavior, frequent lack of etiology, and the lack of scientific evidence regarding various therapies, managing FDB is often challenging. Chronic monitoring and frequent rechecks are often necessary to eliminate the behavior, and sadly owner compliance often diminishes quickly over time, considering the long-lived nature of these birds. The overall prognosis varies significantly as it depends on severity and duration of the behavior—the prognosis is worse for birds with self-mutilation behaviors.
- Knemidokoptes pilae is also called the scaly leg mite, and the cause of “tassel foot” in canaries. Clinical signs of infection include hyperkeratosis and crusting of the cere, beak, legs, and feet and beak overgrowth (Figure 4). Clinical signs are considered pathognomonic, but skin scraping can be performed for definitive diagnosis. The most common treatment is ivermectin, either applied topically, orally, or parenterally for one to three treatments.

Figure 4. Severe overgrowth and a classic “pitted” appearance of the rhinotheca (keratin of the upper beak) and hyperkeratosis of the cere in a budgerigar infected with Knemidokoptes pilae. Photo courtesy Dr. Olivia Petritz Avian and reptilian dermatology share similar disease processes to domestic mammals, including dermatitis and ectoparasites. However, there are numerous diseases unique to these taxa, and these differences are important for veterinary practitioners to recognize.
There are several newly described fungal dermatopathies of lizards, snakes, and turtles requiring specific treatments and can be life-threatening. Feather-destructive behavior in birds is also a common but complex disease, which can become life-threatening if it transitions to self-mutilation.
Olivia A. Petritz, DVM, DACZM, graduated from Purdue University, and then completed several internships and a residency in the field of zoo and exotic animal medicine. Dr. Petritz became a diplomate in the American College of Zoological Medicine in 2013 and specializes in zoological companion animals (exotic pets). Petritz started an exotics service at a specialty hospital in Los Angeles following her residency, and currently is an associate professor of Avian and exotic animal medicine at North Carolina State University.
References
- Abou-Zahr, Tariq. “Avian Dermatology.” Veterinary Clinics: Exotic Animal Practice 26.2 (2023): 327-346.
- Aguilar, Graciela, and Mark A. Mitchell. “Reptile Dermatology.” Veterinary Clinics: Exotic Animal Practice 26.2 (2023): 409-424.
- Sarvi, Jasmine, and David Eshar. “Rodent Dermatology.” Veterinary Clinics: Exotic Animal Practice 26.2 (2023): 383-408.
- White, Stephen D. “Rabbit Dermatology.” Veterinary Clinics: Exotic Animal Practice 26.2 (2023): 347-357.
- Halck, Mette Louise, Nico J. Schoemaker, and Yvonne RA van Zeeland. “Ferret Dermatology.” Veterinary Clinics: Exotic Animal Practice 26.2 (2023): 359-382.
- Eshar, David, and Tali Bdolah-Abram. “Comparison of efficacy, safety, and convenience of selamectin versus ivermectin for treatment of Trixacarus caviae mange in pet guinea pigs (Cavia porcellus).” Journal of the American Veterinary Medical Association 241.8 (2012): 1056-1058.
- Langlois, Isabelle. “Medical causes of feather damaging behavior.” Veterinary Clinics: Exotic Animal Practice 24.1 (2021): 119-152.
- McEntire, Michael S., et al. “Single-dose pharmacokinetics of orally administered terbinafine in bearded dragons (Pogona vitticeps) and the antifungal susceptibility patterns of Nannizziopsis guarroi.” American journal of veterinary research 83.3 (2022): 256-263.
- Mancinelli, E., et al. “Husbandry risk factors associated with hock pododermatitis in UK pet rabbits (Oryctolagus cuniculus).” Veterinary Record 174.17 (2014): 429-429.
- Stelow, Elizabeth. “Avian Behavior Consultation for Exotic Pet Practitioners.” Veterinary Clinics: Exotic Animal Practice 24.1 (2021): 103-117.
- Woodburn, Daniel B., et al. “Emydomyces testavorans, a new genus and species of onygenalean fungus isolated from shell lesions of freshwater aquatic turtles.” Journal of clinical microbiology 57.2 (2019).